Bacterial toxins – a direct cause of pain and inflammation
Researchers in Erlangen discover new approach for developing medication
Pneumonia, meningitis and sepsis are particularly dreaded diseases because they are very difficult to treat. All of these diseases have one thing in common: they can be triggered by gram-negative bacteria, a group of bacteria which includes many types of hospital-acquired infections. Researchers at FAU have now shown that the toxins produced by these bacteria are a direct cause of severe pain and inflammation. These findings could contribute to the development of new medications which could be used to treat many inflammatory diseases.
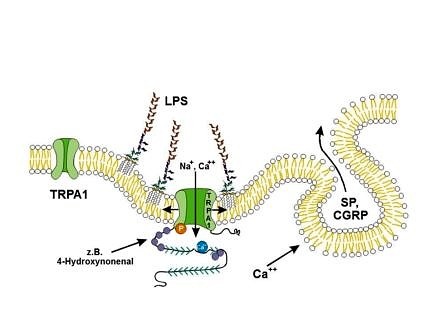
The cell walls of gram-negative bacteria are stabilised by lipopolysaccharides (LPS), chains of sugar molecules which contain fat. When these bacteria are attacked by the immune system or antibiotics, they disintegrate and release large amounts of LPS. The LPS usually act as signalling substances and activate the immune system’s cells. A European team of researchers from Alicante, Leuven and Erlangen, which includes a working group led by Prof. Dr. Peter Reeh from the Institute for Physiology and Pathophysiology at FAU, has discovered that LPS can also be a direct cause of pain. This is because they can activate and sensitise nociceptors – fine nerve branches which can cause the sensation of pain.
These nerve fibres are particularly sensitive due to a common chemoreceptor, the TRPA1 protein, an ion channel which is located in the cell wall. Ions flow in through this channel and stimulate the nerves. This results in a sensation which will be familiar to anyone who has ever chopped an onion – the TRPA1 responds to the irritant released as a gas, causing the unpleasant burning sensation in the eyes and nose.
Receptor reacts to LPS
The team of researchers led by Peter Reeh have shown that LPS can activate TRPA1 quickly and directly, without going via immune cells and inflammatory substances. ‘During our research, we also discovered a previously unknown mechanism,’ Peter Reeh explains. ‘The fat in the LPS molecules appears to penetrate the nerve cell membrane which is also made of fatty substances, causing a dent in it.’ This leads to tension on the TRPA1 which makes it easier for the ion channel to open, allowing sodium and calcium ions to enter and stimulate the fine branches of the nerves. ‘Of course, LPS do not occur alone in diseases; they are always accompanied by acids and other harmful substances which are caused by the bacterial infection,’ says Reeh. The FAU researchers have shown, for example, that when LPS meet these harmful substances at the TRPA1 receptor, highly effective signalling substances called neuropeptides are released by the stimulated nerve fibres, causing blood vessels to expand and the permeability of their walls to increase, while also attracting immune cells. ‘If this happens in several locations in the body, there is a risk that circulation will fail which can in turn lead to endotoxic shock,’ Reeh explains.
TPRA1 blocker could save lives
For the pharmaceutical industry, this effect highlights the importance of developing a medication to stop TRPA1, which has long be suspected to play a harmful role in asthma, colitis and pancreatitis. ‘A TRPA1 blocker might also be able to prevent the often fatal consequences of blood poisoning which can lead to a state of inflammation in the whole body and multiple organ failure,’ says Reeh.
doi:10.1038/ncomms4125
Victor Meseguer, Yeranddy A. Alpizar, Enoch Luis, Sendoa Tajada, Bristol Denlinger, Otto Fajardo, Jan-Albert Manenschijn, Carlos Fernández-Peña, Arturo Talavera, Tatiana Kichko, Belén Navia, Alicia Sánchez, Rosa Señarís, Peter Reeh, María Teresa Pérez-García, José Ramón López-López, Thomas Voets, Carlos Belmonte, Karel Talavera, Félix Viana
Further information:
Prof. Dr. Peter Reeh
Phone: +49 9131 8522228
reeh@physiologie1.uni-erlangen.de
Dr. Tatjana Kichko
Phone: +49 9131 8522692
kichko@physiologie1.uni-erlangen.de