Receptor shown in 3D
Crystal structures could help develop new medications
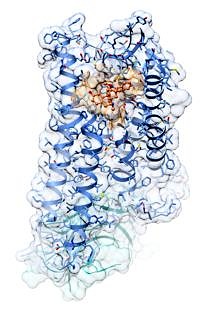
To most people it looks like a cloud with blue streamers and bent straws floating in it. For experts, this image of the crystal structure of an activated μ-opioid receptor – a type of G protein-coupled receptor – could mean a considerable step towards developing strong pain killers that are highly effective and have almost no side effects. An international team of researchers including pharmaceutical chemists from FAU has published images of the three-dimensional molecular structure in the latest issue of the journal Nature. The aim of the working group, led by Nobel Prize winner and Stanford University professor Brian Kobilka, is to develop active agents that are specially designed to activate this receptor.*
Opiates have been one of the most important drugs for thousands of years. They are used to relieve severe and chronic pain, making them incredibly valuable to society. However, due to the feeling of euphoria they can induce and the fact that they are addictive, opiates – which also include heroin – can also have terrible effects and, in the case of an overdose, can cause death due to respiratory arrest. Despite intensive research, no one has yet managed to separate the addictive effects of opiates from their beneficial analgesic properties. Developing pain killers that are free from side effects is therefore still an important goal in pharmaceutical research.
For several years, researchers have been investigating a promising approach known as structure-based drug design. This approach requires detailed knowledge of the exact structure of a particular receptor in order to produce an active agent that docks to the receptor like a puzzle piece. The international team of researchers, which includes Prof. Dr. Peter Gmeiner and Dr. Ralf Kling from the Chair of Medicinal Chemistry, has now succeeded in creating 3D images of the crystal structure of the μ-opioid receptor – the crucial starting point for developing strong opiate pain killers. The high-resolution images of the crystal structure of the μ-opioid receptor that have recently been published in the journal Nature raises hopes that it will be possible to develop new effective and safe pain killers in the future.
A detailed molecular understanding of the interaction between the active agent and receptor will be a valuable starting point for the structure-based drug design of new pain killers. These pain killers will be designed to produce pain-inhibiting signals issued by the receptor by activating the G protein, while blocking signals that are transmitted via the binding of the protein β-arrestin. These kinds of active agents, also known as biased ligands, are described as functionally selective as they trigger the desired effect without any side effects. The concept of functionally selective GPCR ligands is a promising area of not only pain research but also other fields being investigated by the pharmaceutical industry. Prof. Gmeiner’s team is also involved in these research activities. As part of a collaborative project run by the American National Institutes of Health that is also being co-ordinated by Prof. Kobilka from Stanford University, the working group is developing new active agents on the basis of current high-resolution images of the crystal structures of receptors. FAU is also involved in the Research Training Group GRK1910 which is carrying out research on structure-based GPCR drug design.
*doi: 10.1038/nature14886