Patients receive a voice in drug development
Erlangen-based medical researchers participate in European research project with patient preference study
The patient perspective is important in all medical research, and particularly in drug development. This is reflected in the European research initiative Patient Preferences in Benefit-Risk Assessments during the Drug Life Cycle (PREFER) in which a research group led by Dr. Axel Huber at Universitätsklinikum Erlangen’s Department of Medicine 3 is involved. The aim of the initiative, which has received both public and private funding, is to collect information on patients’ preferences and expectations with regard to the benefits and risks of medications for inclusion in guidelines on drug development for industry, regulatory authorities and assessment bodies. The research group based at Universitätsklinikum Erlangen is supporting the initiative by developing and conducting studies of preferences among patients who have or are at risk of developing rheumatoid arthritis.
Drugs are developed for patients and there is an emerging consensus that they should be involved in the identification of benefit-risk considerations during the life cycle of medicinal products. The patient voice is becoming increasingly important, not only for the companies that develop new therapies, but also for the authorities that assess, regulate and decide which drugs are effective, well tolerated and cost effective for patients and the community.
Industry, regulatory authorities, health technology assessment bodies, reimbursement agencies and patient organisations are in agreement regarding the importance of patient preferences. However, there is little guidance on how to conduct studies on this topic that can be used in the pharmaceutical industry, or in decisions on regulations and reimbursement policies. PREFER aims to address this by providing a set of systematic methodologies and recommendations for assessing patient perspectives and including them during the development, approval and monitoring of new therapies.
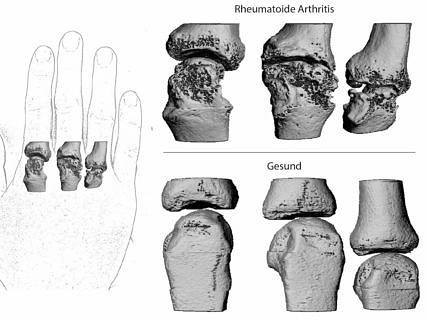
The working group based at Universitätsklinikum Erlangen will contribute to the project by conducting a clinical study of patient preferences among patients who have or are at risk of developing rheumatoid arthritis. Patients with chronic inflammatory rheumatic diseases are usually treated with medication, which is often expensive, throughout their lives. In light of this, it is particularly important that patient’s expectations with regard to the effect and success of medication, as well as their acceptance of potential risks and side effects, are understood. ‘We cannot improve the process of drug development and use without considering patient opinion,’ says Dr. Axel Hueber, head of the working group based at Universitätsklinikum Erlangen. ‘The aim of our project is to test the methodologies and recommendations developed as part of PREFER in a clinical study,’ he continues.
‘Almost all decisions taken in research, trials and approval somehow affect patients, which is why we involve patients directly as partners in the PREFER project. We have also created patient advisory groups to make sure the project gets input from a wider patient community,’ explains project co-ordinator Mats G. Hansson, Professor of Biomedical Ethics at Uppsala University’s Centre for Research Ethics and Bioethics (CRB).
The PREFER project has received six million euros of funding for a period of five years from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement no. 115966. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and the European Federation of Pharmaceutical Industries and Associations (EFPIA). The pharmaceutical industry is matching the amount of financial and human resources provided by the Innovative Medicines Initiative 2. The 33 partners involved in the project include 11 European universities, 16 pharmaceutical companies from Europe and the USA, 4 patient organisations, and 2 small and medium-sized enterprises.
Further information:
Dr. Axel Hueber, PhD
Phone: +49 9131 8539109
E-mail: axel.hueber@uk-erlangen.de