A thin film of air protects materials from liquids and pathogens
Looking to insects to stabilize the surfaces of materials
Several insects use a trick to dive into water without getting wet: tiny structures on their surface trap a thin film of air that protects the animals.
Together with an international team of researchers, Dr. Alexander B. Tesler from the Chair of Corrosion and Surface Technology and Prof. Dr. Wolfgang H. Goldmann from the Chair of Biophysics at FAU have now succeeded in transferring this principle to materials.
In future, these materials’ properties of repelling dirt and microorganisms may lead to significant improvements in the fields of medicine, for instance being used for artificial joints, or engineering, for instance being used for ships’ hulls.
On the way towards this ultimate goal, research groups at FAU, Erlangen, Harvard University in Cambridge, USA, Aalto University in Espoo, Finland, and North Dakota State University in Frago, USA, have now made a major breakthrough, as reported in the journal Nature Materials. Coordinated by the FAU researchers Alexander B. Tesler and Wolfgang H. Goldmann, they have now succeeded in stabilizing surfaces to a far greater degree than has been possible up until now.
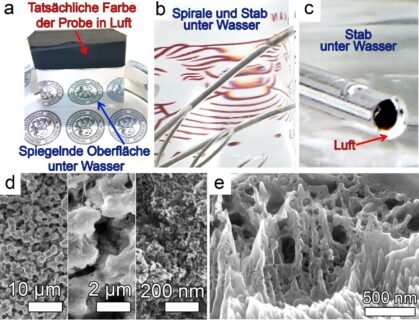
Whilst these materials usually rapidly lose the thin film of air on their surfaces, the protective film remained intact even after 208 days under water during the experiments conducted at FAU. “We suspect that it would remain intact much longer, and would like to demonstrate that this is the case in further studies,” explains Alexander B. Tesler.
For their experiments, the group used an alloy often used in medical engineering applications that consists to 90% of titanium, six percent of aluminum and four percent of vanadium.
The surface of this material is treated with a combination of well-known electrochemical reactions and an alkaline liquid that can be used again and again, making the procedure extremely sustainable. The procedure involves removing part of the surface, leaving an extremely rough surface with individual protrusions of roughly one thousandth millimeter in height. The distance between these structures that are only micrometers in size is in the nanometer range of millionths of millimeters.
Materials treated in this way have surprising properties. If a platelet made of an untreated titanium alloy is dipped in blood just one time and lifted straight out again it is coated in a very obvious red film. However, no visible blood residues adhere to the same alloy when it has a rough surface at the micro- and nanoscale, even after being dipped in blood multiple times.
Another experiment provides an even more fascinating demonstration: whilst drops of blood rapidly leave a smeary red film when they drip off a common aluminum alloy, they shoot off a super rough surface at the speed of lightning without leaving any visible traces of blood.
A similar situation occurs with bacteria and other cells which adhere well to the surface of untreated titanium alloy, but are barely perceptible on platelets that have a rough surface at the micro- and nanoscale. “Microorganisms usually adhere to a solid surface that is hidden beneath the film of air,” explains FAU researcher Wolfgang H. Goldmann. Without a firm base, the tiny organisms cannot survive long.
That is good news for patients who need an artificial hip or knee joint or whose coronary arteries are kept open with stents. If implants such as these are made of standard titanium alloys, patients require treatment with antibiotics to prevent infections. “We hope that antibiotics will become superfluous in future by using the same material with a surface that is rough at the micro- and nanoscale,” states Wolfgang H. Goldmann. Until such a time, however, several more years will be needed in research and development.
The same holds true for use under water. “Air-coated surfaces have not yet played a major role in the development of coatings for ships and other maritime applications,” explains Stefan Kolle. The researcher from Harvard University continues, “the stability of the film of air that we have demonstrated here and an understanding of the fundamental principles open the door to new opportunities for environmentally friendly and low-friction surfaces for use in the sea.”
One important area of application could be, for example, to prevent mussels and barnacles adhering to ships, which can cause ships, ferries and cargo ships to slow down and lead to increased consumption of fuel. With its study, the group has now shown that mussels and barnacles would have virtually no chance to stick to the surface of the titanium alloy with a surface that is rough at the micro-and nano-scale. However, this material would be far too expensive for a ship’s hull. The Harvard researcher Stefan Kolle is convinced that “other methods could be used to create similarly rough surfaces, also using other metals and their alloys.” At the moment, materials such as these are still a long way off. However, the experiments coordinated by FAU have flung the door open wide to such materials becoming available in the future.
Further information
Dr. Alexander B. Tesler
Chair of Corrosion and Surface Technology
alexander.tesler@fau.de
Prof. Dr. Wolfgang H. Goldmann
Chair of Biophysics
wolfgang.goldmann@fau.de